Category: "Photos"
Capturing Falling Snow in a Cold Fluid
February 15th, 2021Snow is usually imaged in air, the single crystals laying flat on some substrate such as glass. The method is relatively simple, but one must work fast to image the crystal before it appreciably sublimates. Sublimation first rounds out the sharp edges and then causes the crystal to shrink. Generally, this sublimation happens because the photographer is radiating too much heat to the crystal. Conversely, particularly in very cold conditions, the photographer’s breath may deposit fog near the crystal, causing the crystal to grow.
Such issues vanish if one instead captures the snow in a cold fluid before taking the image. To work, this fluid should not dissolve the ice, be less dense than ice, be fluid enough to completely spread over the crystal surface, and be transparent. Other than preserving the crystal, the method has several other advantages. For example, in his laboratory experiments in Hokkaido, Japan, Tsuneya Takahashi lets the crystal fall into a cold suspension of two transparent, cold silicone oils. He sets it up so one fluid is denser than ice, one is lighter than ice, so the crystal falls through the light oil and rests on the (transparent) interface between the two fluids.
This method sounds complicated, so why use it? One, as the oils are immiscible with water, they block water molecules from arriving or leaving the crystal surfaces, so the ice crystal shape is preserved precisely for as long as the fluid is below 32 F (0 C). Two, after imaging the crystal, the fluid is warmed above melting such that the crystal melts into a spherical drop from which he can easily measure the volume and thus infer the mass of the original crystal. A third advantage, more difficult to exploit yet sometimes used, is that he can get top and side views of the same crystal. Other researchers in Japan have also used silicone oils to capture ice crystals in the lab, as well as naturally falling crystals, mainly for the first and third reason. They use just one oil type, a lighter oil. Charles Knight at NCAR in Boulder, Colorado had a fourth reason for using a cold fluid: better imaging. That is, one can image greater depth detail because light scattering off the surfaces is greatly reduced, particularly if the fluid is very clear and has an index of refraction close to that of ice. By reducing the scattering, one can see through surfaces to underlying surfaces. He would use gasoline or hexane fluid.
I don’t have a photomicrography setup to take detailed images of snow, and we rarely get snowfall with nice single crystals anyway, but I wondered how well the method might capture falling snowflakes. That is, could I at least see their rough shapes as they fell through the fluid?
Here, we typically get about one light snowfall per winter (2-3"). A relatively large snowfall happened this past weekend, depositing about seven inches. At first, the particles were small, probably highly rimed single crystals or small aggregates. Later, larger snow particles fell, and these particles were clearly snowflakes (i.e., aggregates). In preparation, the previous night I set out two covered wide-mount jars, one with water, the other with Coleman camping fuel (white gas), which has similar properties to gasoline. In the morning, the one with water had frozen, so I knew the other was also below 32 F.
Outdoors, I set the jar on top of my car, put a wooden chopstick in the jar both to focus on (my camera only has autofocus) and to provide a size reference, set a small LED light panel to the side for brighter illumination, and then opened the lid. The flakes fell into the fluid, and fell down to the bottom of the jar. They fell through the fluid slower than they fell through the air, but it was still too fast for me to see how well they were focused. Turned out that they were not very sharply focused, yet one can still see their general shape and fall orientation (below). In general, the flatter the flake, the more it tends to orient broadside to the fall direction.
Obvious improvements would be a better jar, such as one with a flat, smooth glass front, a better camera, and a thicker fluid to slow down the rate of fall.
Such improvements will have to wait at least until next winter.
--JN
A Stroll on a Mildly Frosty Morning
December 23rd, 2020So far this winter, we've had few frost days in the Redmond, WA area. This morning was typical of the half-dozen or so: a very light dusting of hoar crystals on the roofs and grass. One cannot expect much, and yet I am rarely disappointed. Indeed, it is not until I get close to some icy thing do I notice anything interesting. Sometimes, I still don't notice until I've clicked a few closeups and then viewed on a large computer screen. Here's what I found on this morning's stroll:
The frozen puddle showed some curvy meniscus lines and some straight ice blades, which I've discussed before. The film frost is more mysterious still, but the main curvy pattern is due to the freezing of melt (not vapor deposition, though some of this does occur). The hoar frost shows some scroll crystals, which I recently addressed in an article (no mechanism had previously been argued, but we propose an explanation). See the crystals on the leaf at upper right for the best examples of scrolls, though the details are yet a bit too small. The needle ice is the phenomenon responsible for the crunchy dirt and pushes ice up from the bottom. The ground is warmer below the surface, and here the liquid migrates to the ice front, pushing it all skyward.
Anyway, that's it for this post. No detailed explanations of anything. If you would like to see such explanations, click on the appropriate category in the archives at the right side.
And here's hoping you have a nice, frosty Christmas, wherever you are-
--JN
A rare heliac arc? (plus six others)
February 5th, 2019The day starts sunny and bright, but later you note a slight muting of the surrounding landscape. It is still bright enough, but you feel less heat bearing down. Apparently, a thin veil of high clouds has slowly and silently appeared above.
When this happens, please take a few seconds to look up and see what these clouds are up to. Do they have their crystals lined up for one or more arcs, or are they oriented randomly, giving a halo with perhaps a sun dog or two? To the trained eye, the chances are high that such a veil will give rewards, even when no crystals are present. But the crystals give so much more, particularly when they line up in various ways.
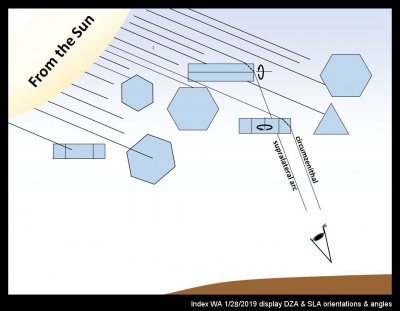
Wandering, somewhat exhausted, on the hillside above Index, WA last Monday, I got this very sense of a muted landscape. So, I found a break in the trees, looked up, and instantly felt recharged. A colorful circumzenithal arc had appeared, which by itself is always a treat. But I was also awed by several other arcs and a very diffuse 22-degree halo. Excitedly, I took photos and scrambled around trees and rock for a more complete view.
(As with all images here, click on the image to see a larger view.)
Due to the complex way the eye and brain discerns light and patterns versus the much simpler way of a camera, the patterns are much more distinct when viewed direct by eye. But the above composition has my attempt to compensate the original photo at left, with some contrast enhancement in the middle version, and markings on the right version.
The band of color at the intersection of CZA and SLA is from the oriented prism crystals of the circumzenithal arc (CZA). Their formation is relatively common I think, though a group of observers in Germany apparently finds their occurrence there to be only about 13 times per year (https://www.atoptics.co.uk/halo/whyinfr.htm). What is most remarkable to me is not their colors, but the fact that formation of distinct colors requires that the crystals stay extremely level as they fall (deviations of only a few degrees would wash out the colors). My sketch below shows roughly what is going on here:
(Hold on for a moment, and I'll get to the rare heliac arc below.)
Columns!
January 18th, 2017The poor columns get left out of nearly all snow-crystal discussions, but they are an interesting type. So, to help them out a bit, here's my first column appreciation post.
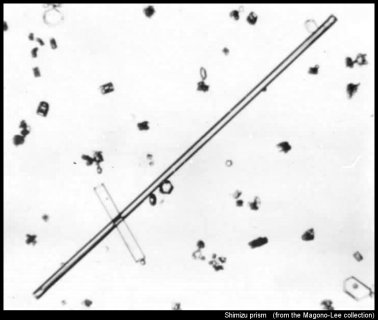
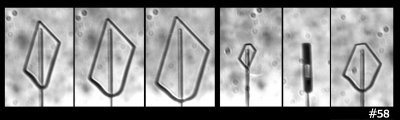
Let's start with perhaps the most extreme column of all, the Shimizu prism*:
I say 'extreme' because they are so long and thin--sometimes over 1-mm long yet just 0.01-0.02 mm in diameter. These types have so far been found to fall only on the Antarctic Plateau. But in theory, they should be able to form elsewhere. It is like a "whisker" crystal, which Teisaku Kobayashi grew below -50 C on a surface in the lab. The image above shows many other crystals as well, including another solid column crossing the Shimizu prism.
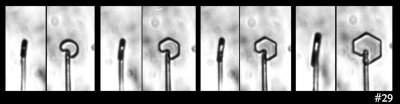
Next, the bullet rosette:
The bullet rosette is most often found below -25 C in high cirrus clouds. It is an example of a polycrystal; in this case, a frozen droplet that froze into several distinct crystals (one for each "bullet").
Next, one of my favorites, the scroll column (though the picture doesn't quite do it justice):
In this form, the sides of the crystal seem to fold inward, like a scroll.
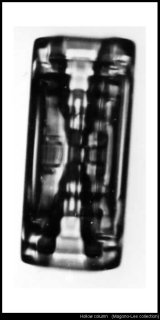
Finally (for now, anyway), the ubiquitous hollow column:
The funny banding you see (horizontal lines inside the 'hollow') is a mystery.
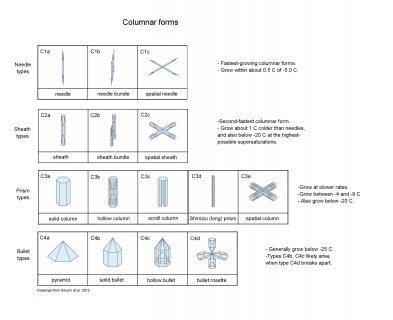
There are many other columnar forms, many of which are in the following diagram (as with all images here, click on it to see it enlarged)**:
One neat thing about the columnar forms is that you can see roughly exact replicas of them in hoarfrost. The Shimizu prism may be hard to find, but the others are common if you look closely.
-JN
* Images are from the Magono & Lee collection, used in their paper: Meteorological classification of natural snow crystals. J. Fac. Sci., Hokkaido Univ., Ser. VII 4, 321–335.
**Drawings are based on those in Kikuchi, Kameda, Higuchi, and Yamashita: A global classification of snow crystals, ice crystals, and solid precipitation based on observations from middle latitudes to polar regions. Atmos. Res. 132-133 (2013) 460-472.
More frost patterns on black cars
January 7th, 2017Black cars remain my favorite place to observe frost patterns. Here are a few I saw on one car today.
The straight lines of frost are more common when it is drier. And it has been quite dry here due to the very low temperatures.
These pictures show an interesting mixture of straight lines and curved boundaries.
And finally, the windshield had a pattern that resembled a hilly landscape.
But the pattern is actually quite flat. The frost is playing mind games on us, presenting an optical illusion of 3D topography.
As with all cases of frost on surfaces, the ice initially got started when a thin layer of liquid (melt) froze in various spots. The ice that grew, first grew in the melt layer, then grew on top, essentially "sucking"** the vapor out of the surrounding air, thus drying out surrounding regions. This is why we see bare surfaces near the larger frost crystals. Those frost crystals grew from the vapor, just like snow, but are anchored to the surface because that's where the film froze. So, two types of crystallization are important: freezing of the melt (melt --> ice) then vapor deposition (vapor --> ice).
- JN
** The actual process is diffusion (the way perfume molecules reach our noses), but this term is a little more vivid.
Akira's Corner Pockets
May 10th, 2016The evanescent snow crystal
appears out of nowhere
The lines and boundaries
on its faces record a story
a story of a crystal's birth
a story of a crystal's life
But before the record vanishes
Who will hear its story?
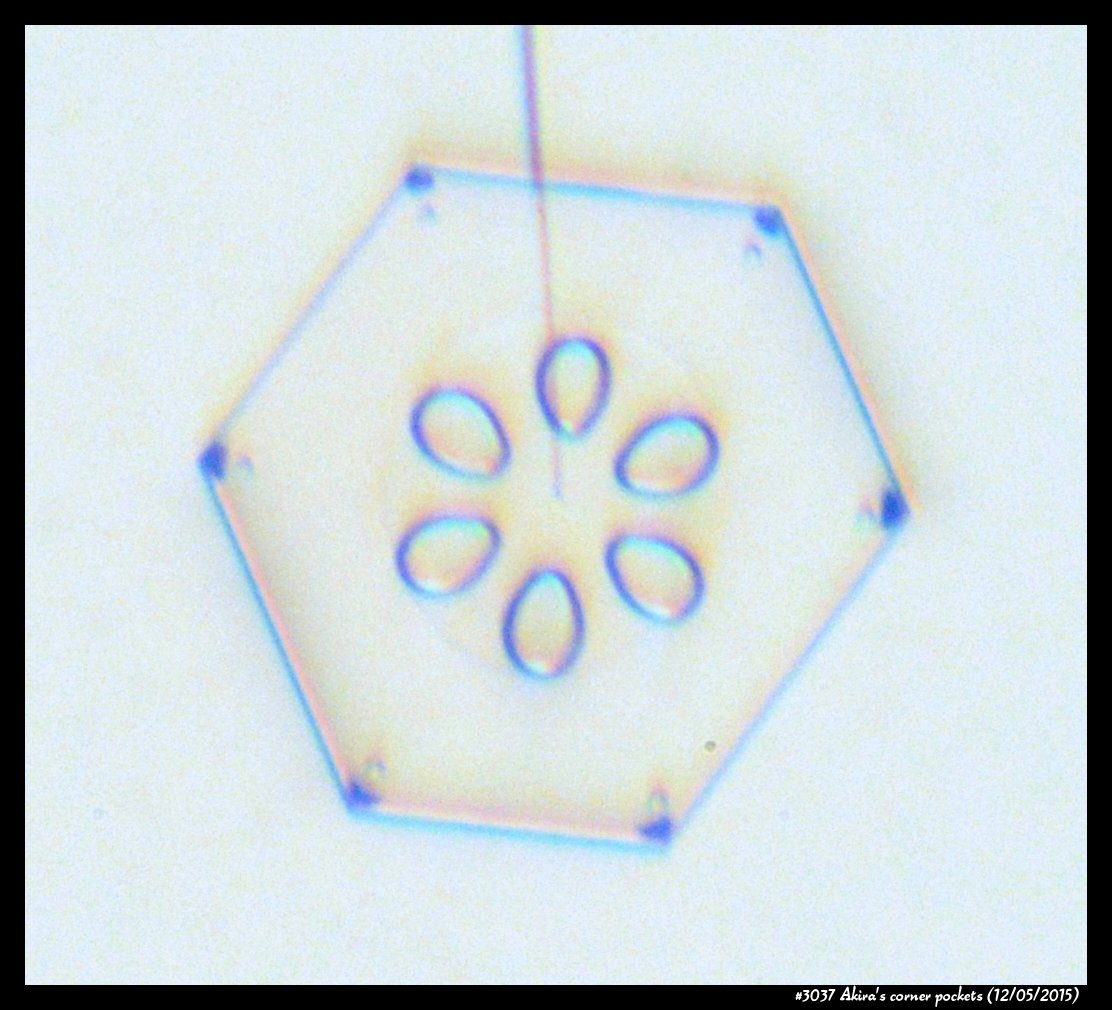
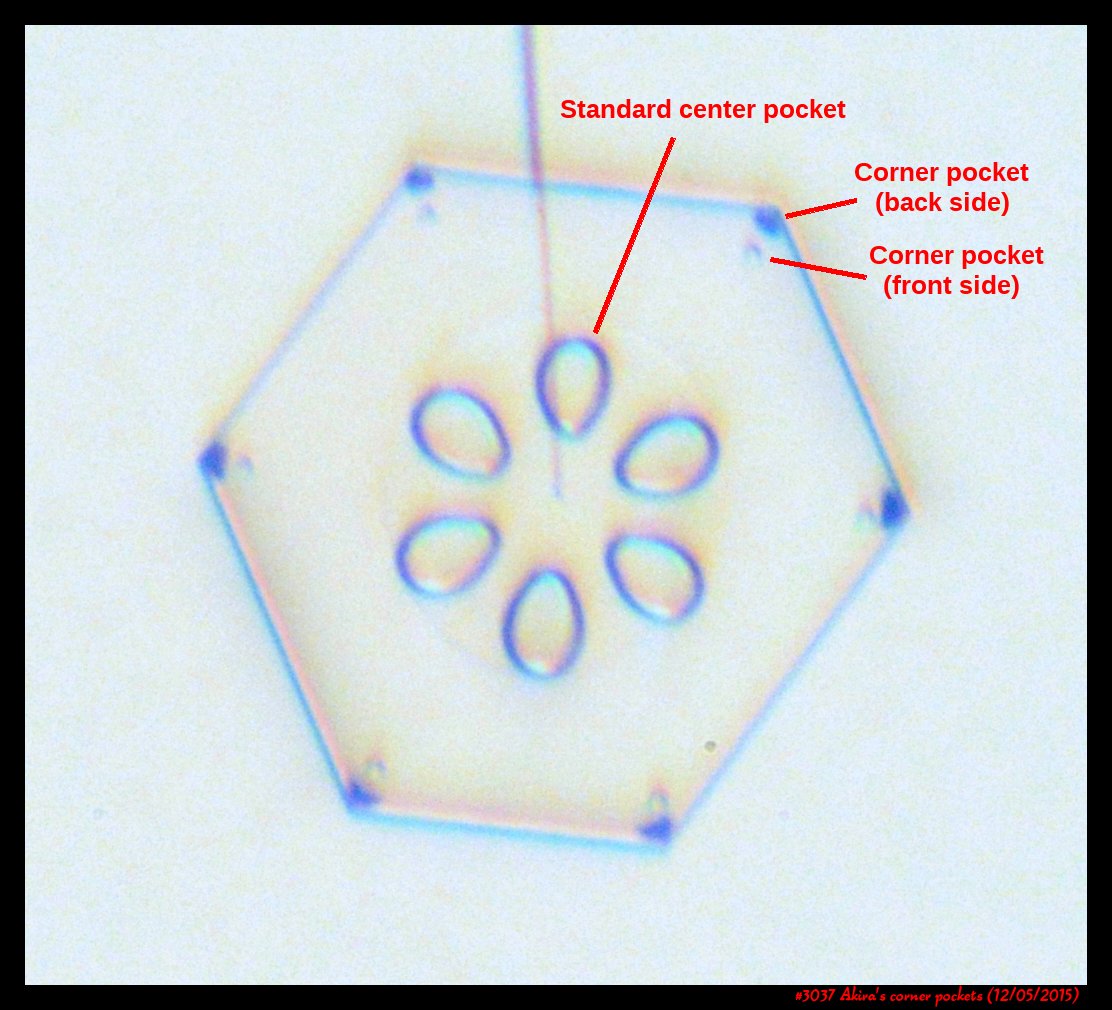
A few years back, a correspondent of mine, Professor Akira Yamashita of Japan, long retired, sends me an email. In the email, he had a document with words and pictures of some small crystals that he'd captured back in the 70s. They were small crystals, essentially freshly "hatched eggs" from the frozen droplets upon from they had started. But some had small pockets of air near their corners.
To those who have studied any sort of crystal growth and have some familiarity with crystal-growth theory, these corner air pockets, or "bubbles", were in impossible locations. They should not be there. Pockets will form near face centers, not corners. But Prof. Yamashita also had a theory about their formation. His theory first looked sketchy to me, but I appreciate hearing about new ideas, so over the following years kept revisiting his theory, getting to think that it had merit, and wondering if it had other applications.
Then, just this past year, in our own ice-crystal experiments, we did something that apparently had never been done to small ice crystals in the lab before. We slowly grew a crystal in air. And we cycled it from slightly growing, to slightly sublimating (i.e., shrinking in size), to slightly growing again. A cycle that must happen in some regions of cloud. And here is what we saw:
Corner pockets!
After the sublimating, the subsequent growth kept a permanent record of the sublimation cycle in the form of 12 corner pockets, one pocket for each of the 12 corners of the crystal. These are pockets of air, just like the six large 'petal-shaped' pockets of air you see nearer the center of the crystal. They are forever stuck in the crystal. Stuck there until the crystal, with all of its features, vanishes back to air.
After seeing this, we ran a few more experiments, and each time we slowly grew, then sublimated, then grew again, we got corner pockets. The name 'corner pockets' refers to their location when they are formed; namely at the corners, next to the crystal perimeter. However, they remain essentially fixed in position as the crystal grows, and this means that as the crystal perimeter expands outward, the corner pockets will appear further within the crystal. Analogously, the 'center pockets' shown above formed at the face centers, on the crystal perimeter, back when the crystal was much smaller.
As to the theory of their formation, and how the theory can explain other observable features of snow crystals, you'll have to wait for a subsequent post.
- Jon
It’s not a rainbow (it’s better)
August 12th, 2014In summer, the sun reaches higher elevations, bringing the possibility of new atmospheric displays. I saw this one in mid-June.
The top, upward arc is the more familiar and common 22-degree halo. But pay attention to the one on the bottom. Its colors look like a rainbow, but it has nothing to do with water drops of any sort.
It is the circumhorizontal arc, and it is as rare as it is beautiful. The colors are, in fact, more pure than those of the rainbow, and its origin is far more remarkable. It is remarkable because of the surprising set of conditions that must hold for it to exist. First of all, the particular region of cloud (at a certain angle to our eye) must consist mostly of ice crystals. Second, the crystals there must be nearly perfect tabular prisms. And third, these crystals must be in sufficiently non-turbulent air and of such a size that they fall in a nearly exact horizontal position:
Halo in the sky? Uh, I don't see no halo...
April 20th, 2014After a few days of fine bright spring weather, the barometer falls and a south wind begins to blow. High clouds, fragile and feathery, rise out of the west, the sky gradually becomes milky white, made opalescent by veils of cirro-stratus. The sun seems to shine through ground glass, its outline no longer sharp, but merging into its surroundings. There is a peculiar, uncertain light over the landscape; I 'feel' that there must be a halo round the sun!
And as a rule, I am right.
The quote, from Minneart* describes a common ice-related atmospheric apparition. It appears in skies all over the world far more often than the rainbow, yet few notice it. As a graduate student, I read about halos and often looked for one, but didn't notice it myself until someone else pointed it out. As a post-doc in Boulder, I was out walking with Charles Knight, and I mentioned my lack of success. He glanced up near the sun, pointed, and said “why there's one right now”.
What I had missed in my readings had been the fact that most halos are rather indistinct and often incomplete circles. Indeed, now when I point out the most common one (the 22-degree halo) to someone nearby, they often don't see it. But occasionally, it is sharp enough (and colored) to the extent that anyone will see it if they bother to look up and glance toward the sun. And often it occurs with other ice-crystal apparitions that are even more obvious.
Last fall, while perched high on a rock face, belaying my partner up**, I saw such a vivid display.
The bright spot is called a “sun dog”, “mock sun”, or “parhelia”. They, one on either side of the sun, usually appear together with the 22 degree halo. Indeed, the sun dogs very nearly mark the spots where that halo intersects another arc called the parahelic circle. Their cause: horizontally oriented, tabular ice crystals.
The fun of shooting down your own theories
April 10th, 2014Thomas H. Huxley once wrote the famous line:
The great tragedy of Science: the slaying of a beautiful hypothesis by an ugly fact.
Great man, catchy phrase, but perhaps a bit overdramatic. To me, the slaying of a “hypothesis” (i.e., pet theory or just idea, really) can be the beautiful thing. It means that one can do a lot of damage with just a simple observation. I like it, even when I'm shooting down my own theory. Here's an example:
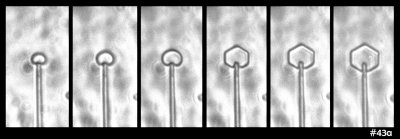
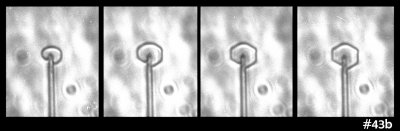
Some time ago in my experiments, I saw the following ice growth sequence:
What you see there is an extremely small, thin ice crystal growing from the tip of a glass capillary into air. (Size-wise, the glass capillary is about 5 micro-meters in diameter, or about 1/10th the thickness of the hair on your head.) I saw it happen several times. As the crystal grew, it developed the prism facets that generally define the hexagonal crystal shape. Other people had seen such rounded growth before, generally within a few degrees of zero (C), though in all cases, the crystal had been extremely thin. You can also see this thin, rounded (non-facetted) form in some hoar-frost formations.
The mystery here is why the disk grows without the prism facets for awhile. I never saw this with thicker crystals, so I formed a little theory. The theory involves the source of the water molecules to the curved region of crystal: some come from the vapor in the air and some wander over from the flat, non-growing crystal faces on front and back. In the 1960s, people had tried to measure this “wandering distance”, but never determined a consistent value. My theory predicted that once the crystal thickness exceeded about twice this distance, the curved edge would transition to flat, giving rise to the hexagon.
But then I looked closely at this case:
That sequence shows the side and front view. The crystal doesn't discernibly thicken when the flat prism facets appear. Zing! That theory shot down.
On to the next pet theory. So maybe the key factor is the diameter of the disk: One might argue that the curvature of the crystal surface must be below a certain value, which means a large enough diameter is needed, for the flat facets to appear. Or, the size of the resulting prism faces must be larger than that needed to have several surface steps (which help ensure flatness).
But then I look at this case:
In that case, I see both smaller and larger prism faces forming at the same time (same thing can be seen in the previous image). So, I guess the curvature or size of the resulting face is not the main factor. Zing!
Some researchers had observed slight bending of the prism facet above -2.0 C in equilibrium. They postulated a “roughening temperature” at -2.0 C. This might explain the rounded disc edge, but wait! 1) This disc edge becomes facet as it grows, so it is not merely temperature, and 2) these crystals are below -2.0 C.
Zing again!
Well, there are always “impurities” to blame! Crystal growers are fond of blaming some trace, active chemical, or “impurity” for inexplicable results, so we could theorize that the above show the effect of surface impurities. As the crystal grows, the area over which the impurities distribute increases, thus diluting their concentration, and thus reducing their effect. Yes! This could explain these results.
But, wait, what about this:
The above shows two sequences (same capillary) in which some prism facets have formed, but some remain round. In the right-side case, one corner even rounds as it grows. Hmm, not a likely result of impurities. Zing!
So, I am down to one last theory. I do not yet have the data to shoot it down. And I'm not telling you, or I'd ruin the fun. We just need more data.
All in all, I think Huxley needs a little tweaking to serve my view:
The great beauty of science: the slaying of a pet idea by a simple observation.
-JN
Equinox frost I: Patterns on roof, hood, window, and mirror
March 24th, 2014Morning on the spring equinox, the first day of spring, brought a few gifts from winter. I) Film-frost, accentuated with hoar-frost on the cars.
See the white of the hoar, following a pattern set down by the thin layer of film-frost. The roof of one car:
And the roof of another:
So it went. As I walked through our parking area, I saw a different pattern on every car - a pattern that told a tale of the night's weather and evening conditions. The film frost here is dramatic because the water film was thick before it froze. And it was thick because it had been raining in the previous evening. Later that evening, the skies cleared, and the temperature dropped rapidly, ensuring that the thick film would freeze. And the warm wet weather of the previous day left plenty of vapor to deposit as hoar-frost.
Car hoods had their own story to tell: